The central atom should hopefully now have a full outer shell of electrons.Add any remaining electrons to the central atom.Once again, remember to count any bonds as pairs of electrons. This will give you your number of remaining electrons. Count up how many electrons you've added to your molecule in total, and subtract this from the molecule's number of valence electrons that you worked out earlier.
Remember to count any bonds as a pair of electrons.

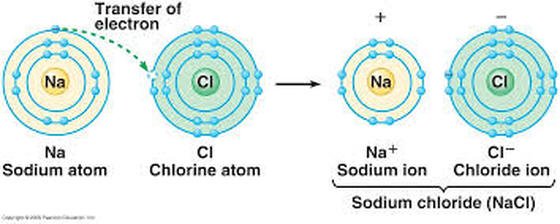
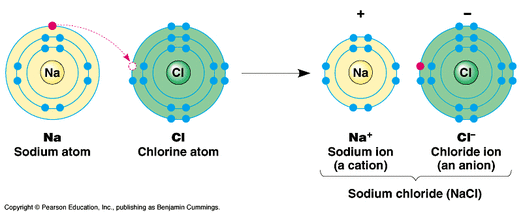
This usually means that they need eight electrons, but there are some exceptions.

We can also draw Lewis dot diagrams for uncombined atoms that have yet to form a molecule. Anna Brewer, StudySmarter Originals Lewis dot diagram for carbon Here is another example: the Lewis dot diagram for methane, CH 4.Ī Lewis dot diagram for methane. Electrons are generally spaced out evenly around all four sides of the atom, with two per side.Atoms are at their most stable when they have full outer shells and the octet rule states that for most atoms, this occurs when they have eight valence electrons. Lewis dot diagrams are based on the octet rule.A single line represents a single covalent bond with just one bonded pair of electrons, whilst two lines represent a double covalent bond with two bonded pairs of electrons. Bonded pairs of electrons are shown as lines, signifying a covalent bond.Two dots side by side represent a lone pair of electrons. Specifically, they show any bonded pairs or lone pairs of electrons. They also show the number, position and arrangement of the atoms' valence electrons, whilst assuming that all inner electron shells are complete. Lewis dot diagrams show the atoms in a molecule, be it a neutral compound or a charged ion. Don't worry if you see any of these terms floating around - they all mean the same thing! Lewis dot diagrams are also known as Lewis structures, Lewis dot structures or electron dot structures. Finally, we'll consider the importance of Lewis dot diagrams.Ībove, we introduced you to the idea of a Lewis dot diagram: a simplified representation of a molecule's valence electrons.
#Cl ion bonding properties how to
